Introduction
Clinical stroke after TAVR is frequent (4-7% at 30 days in RCTs), generally under-reported (17% after SAVR when evaluated by neurologist), confers 3- to 9-fold increased risk of mortality and 50% occurs peri-procedural. Silent stoke affects 58-100% of patients and are associated with neurocognitive decline, dementia and stroke. 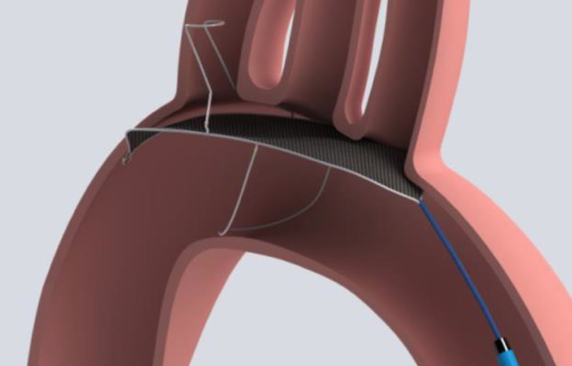
The safety, efficacy and performance of TriGuard (a Single-wire nitinol device for neurological protection positioned across all 3 cerebral vessels) protection compared with unprotected TAVR is assessed in DEFLECT III tiral.
Material and methods
Multicenter prospective single-blind randomized controlled trial at 13 sites, including 86 patients (exploratory study for the design of a pivotal RCT). Primary Safety Endpoint (ITT population): In-hospital composite of death, stroke, life-threatening or disabling bleeding, AKI (2/3), and major vascular complications (VARC-2). Secondary Performance outcome:
successful positioning (complete 3-vessel coverage verified by angiography) and retrieval without TAVR interference. Secondary Efficacy: Frequency, number, and average single, maximal, and total lesion volumes by DW-MRI at 4 days (range 2-6 days). Neurocognitive assessment was performed by Montreal Cognitive Assessment scores.
Results
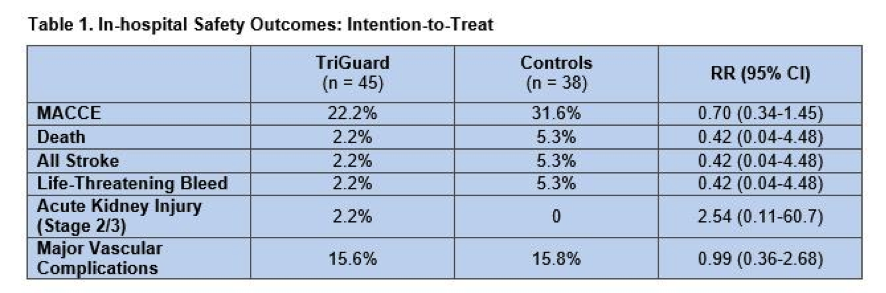
There were not important baseline characteristic differences between both groups. CoreValve (Medtronic) was employed in about a third of patients, with a Sapien device (Edwards LifeSciences) being used in the rest. Fluoroscopy time was longer with the TriGuard (28.2 vs 18.6 minutes; P < .001). 29% and 34% of study and control patients, respectively, were lost to follow-up over 30 days for reasons including stroke, refusal, permanent pacemaker implantation, or death. Technical success was achieved in 87% of the study group (6 patients lost complete coverage), and TAVR success did not differ between the study arms. The primary safety endpoint and its individual components were comparable between both groups (table 1).
At 30 days, 73% of the TriGuard group and 88% of controls had new lesions on diffusion-weighted MRI (4.5 and 4.0 new lesions per patient, respectively). TriGuard reduced single and maximum lesion volumes: 16% and 17%, respectively, in the intention-to-treat analysis, and 41% and 45% in the per-treatment analysis. Within the study arm, the percentage of patients with zero total lesion volume at 30 days was 21.9% in the intention-to-treat analysis and 26.9% in the per-treatment analysis. Only 12.5% of controls had zero total lesion volume. NIH stroke scale score, improved or stayed the same at 30 days for the majority of patients in both the study (97.4%) and control arms (87.9%). Montreal Cognitive Assessment scores improved in the TriGuard arm and worsened in the control arm.
Conclusions
In this exploratory clinical trial, use of the TriGuard was safe and provided complete cerebral coverage in 87% of cases, it increased the proportion of patients completely free of ischemic brain lesions and it reduced single and maximum lesion volume.






