Introduction
Degenerative MR is common, affecting ~600,000 persons in the U.S. Surgery is the standard of care, and is indicated for patients with symptoms or LV dysfunction. However, there are patients in whom the risk of surgery is prohibitive. The Mitraclip system received commercial approval in October 24, 201, being indicated for symptomatic patients with primary MR ≥3 and prohibitive surgical risk. Our objective was to analyze and report the initial commercial experience with the MitraClip System in the U.S.
Material and methods
Figure: Mitraclip device
This study shows all commercial transcatheter mitral valve repair (TMVR) cases with MitraClip (figure 1) enrolled in the registry through August 31, 2014 for 564 participants across 61 health care centers in the U.S. The in-hospital and 30-day outcomes for procedure success, complications, and device-related events, were examined. Procedure success was considered if post-implant MR grade ≤2, without CV surgery and without in-hospital mortality. Other definitions were procedure complications (cardiac perforation, major bleeding, stroke, MI, mitral injury, or death) and device-related adverse events (single leaflet device attachment, complete clip detachment, device thrombosis, device or delivery component embolization)
Results
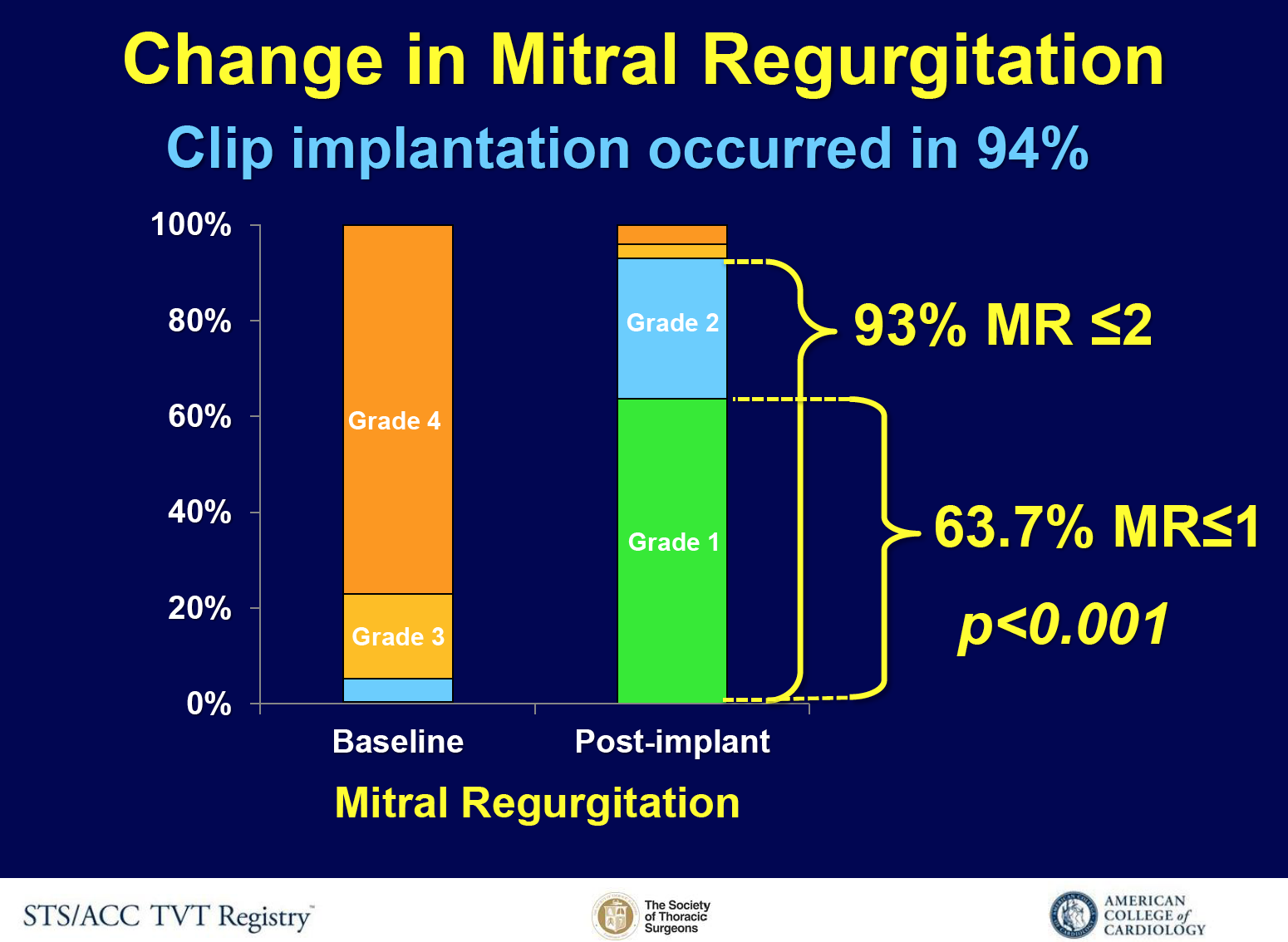
Figure 2: Change in MR
Clip implantation occurred in 94% of the cases (figure 2), and procedural success was achieved in 91.8%. Variables related with procedural success were: smaller left ventricular end-diastolic diameters, less severe baseline mitral regurgitation, A2-P2 clip location and case volume (per 2). Procedure complications and device-related adverse events rates were 7.8% and 2.7%. Mortality was 2.3% in-hospital, and 5.8% at 30-days.
Conclusions
- In this first report of the U.S. commercial experience with TMVR, procedure success, clinical outcomes, and adverse events were favourable in comparison to pre-approval studies and other national registries.
- These data demonstrate effectiveness and safety of TMVR with MitraClip for the treatment of prohibitive risk patients with symptomatic MR






