Introduction
The SAPIEN 3 TAVR system is the newest iteration of balloon-expandable valve designed with an external fabric skirt to minimize paravalvular regurgitation (PVR). Two non-randomized registries were embedded in the PARTNER II trial to evaluate valve performance.
Material and methods
From October 2013 to September 2014, 1661 patients with aortic stenosis [583 high-risk or inoperable (S3-HR), 1078 intermediate risk (S3-I)] were treated via the transfemoral (TF; n=1450, 87%), transapical or transaortic (n=211, 13%) access route at 57 sites. Procedural outcomes and 30-day clinical events were adjudicated by an independent clinical events committee. Echocardiographic results were analyzed by a core laboratory.
Results
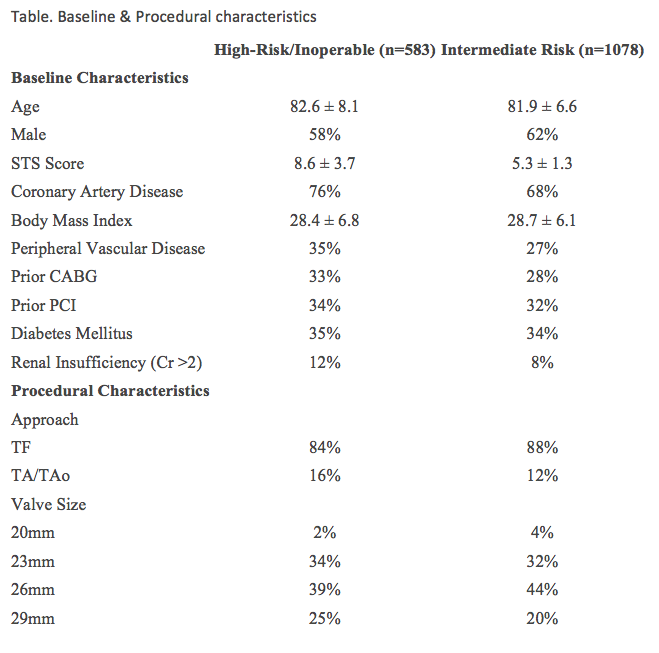
Baseline and procedural characteristics of both cohorts are below (table). In the S3-HR cohort, 30 day all-cause mortality was 2.2% (TF=1.6%); cardiac mortality was 1.4% (TF=1.0%). Other complications included all stroke 1.6%, myocardial infarction 0.5%, major vascular complications 5%, and requirement for permanent pacemaker 13.1%. PVR was none/trace in 61.6% of patients, mild in 35.5%, and moderate in 2.9%. No patients had severe PVR. Mean gradients decreased from 45.5 mmHg at baseline to 11.1 mmHg.
Conclusions
The SAPIEN 3 TAVR system was associated with low 30-day complications and reduced PVR (compared with previous transcatheter valves).






