Introduction
The randomized CoreValve trial demonstrated that transcatheter aortic valve replacement (TAVR) resulted in significantly lower mortality compared with surgical AVR at 1 year in patients who were at increased risk for surgery. Longer-term outcomes following TAVR with the self- expanding CoreValve are necessary to further validate this survival advantage.
Material and methods
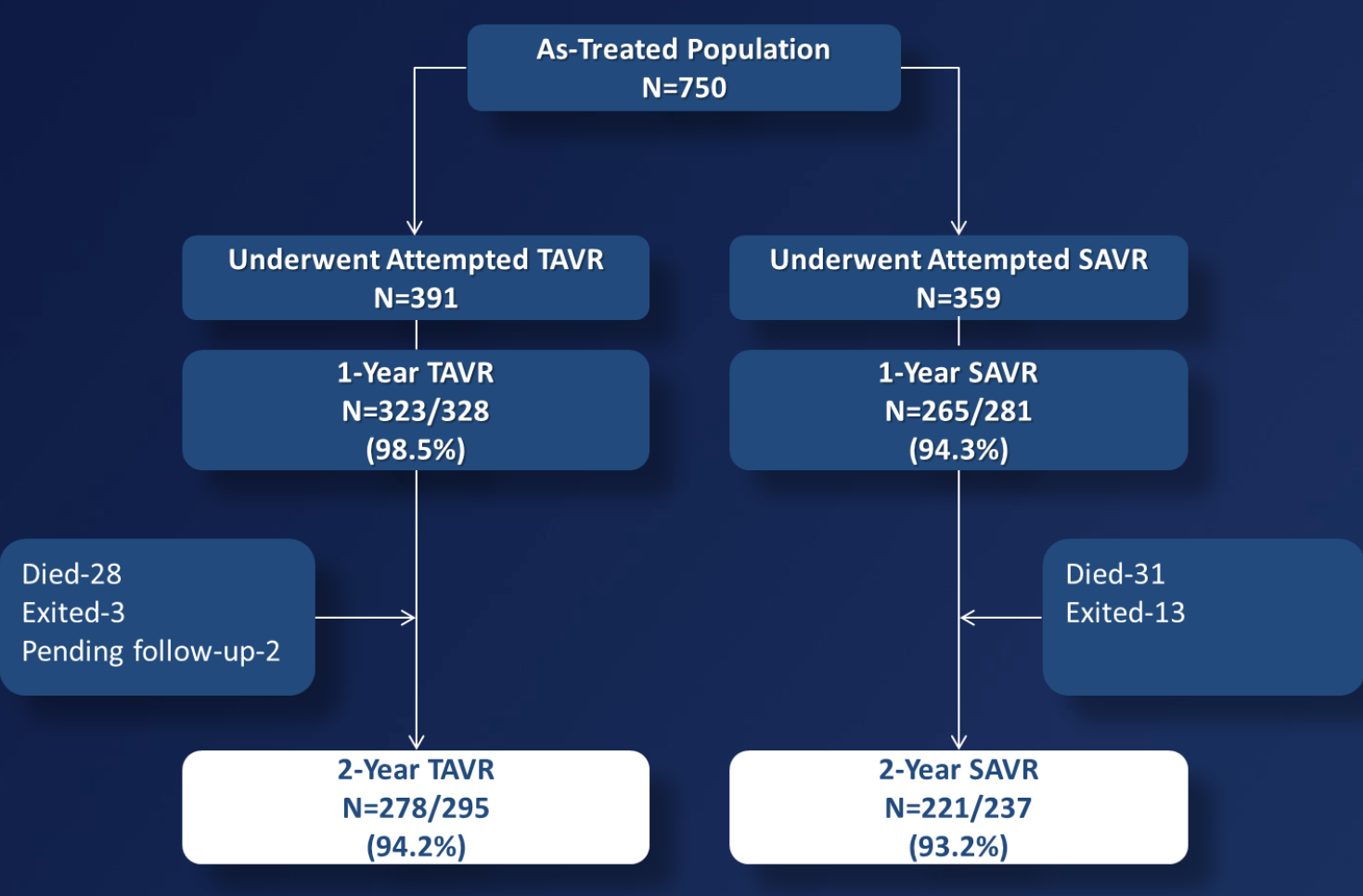
We included patients with severe aortic stenosis at increased surgical risk with NYHA functional class II or greater. The primary endpoint was all-cause mortality at 1 year. Non-inferiority Testing: TAVR with the CoreValve bioprosthesis was non-inferior to SAVR for 1 year all-cause mortality with a 7.5% non-inferiority margin. Superiority Testing: If the primary endpoint was met at the 1- sided 0.05 level, a subsequent test for superiority was performed at the 1-sided 0.05 level
Results
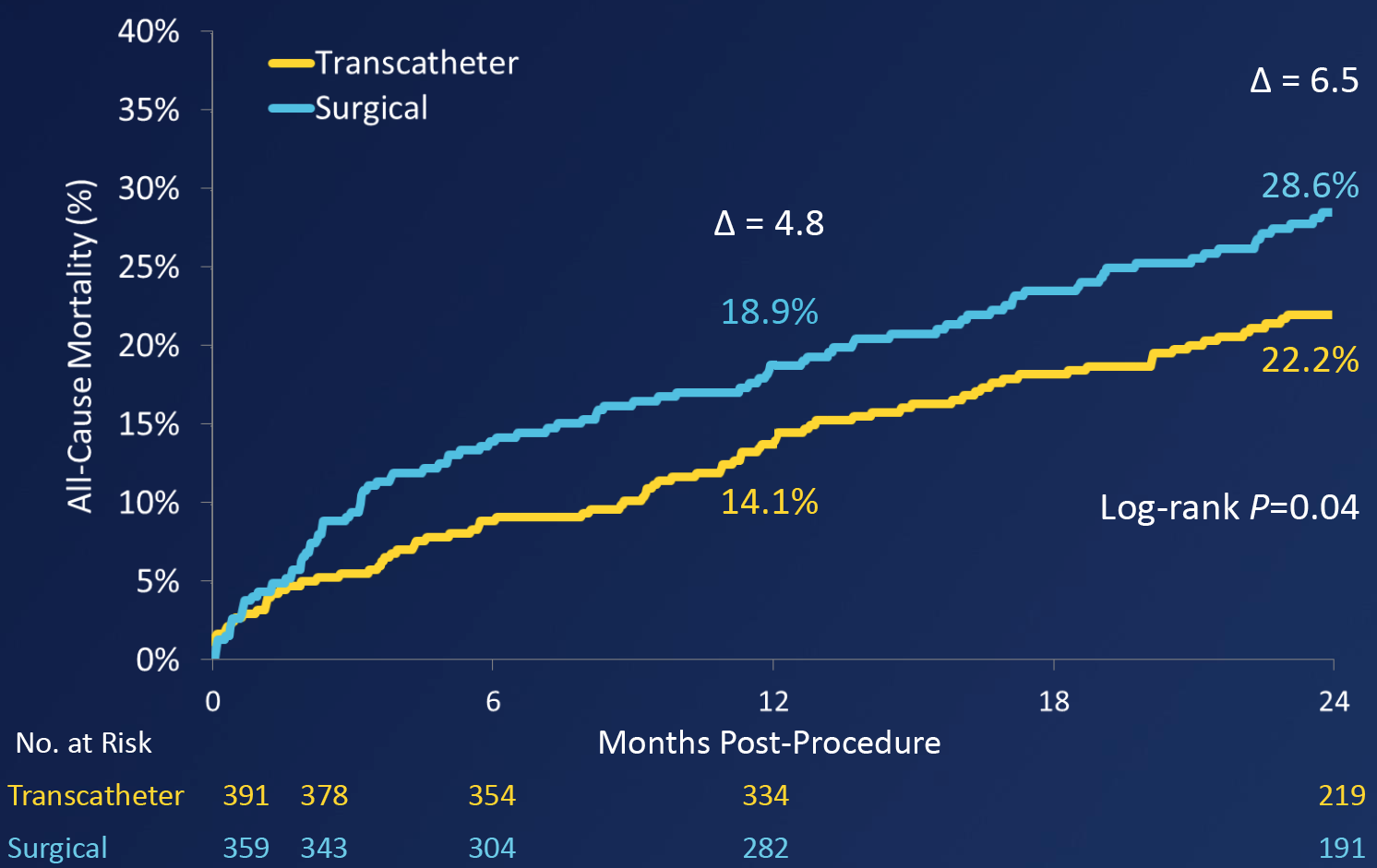
All cause mortality was lower in the TAVI group compared to the surgical SAVR group (22.2% vs 28.6%, Log-rank P=0.04).
Conclusions
At 2 years for patients with symptomatic severe AS at increased risk of surgery, the superior survival seen at 1 year for TAVR over SAVR is maintained. All stroke was less with TAVR over SAVR but major stroke showed no difference and MACCE was significantly lower with TAVR compared to SAVR.






